
The scale is called the Pauling Scale, named after Linus Pauling who created the scale in 1932. There is an electronegativity scale that reflects how strong the bond energies for atoms are. 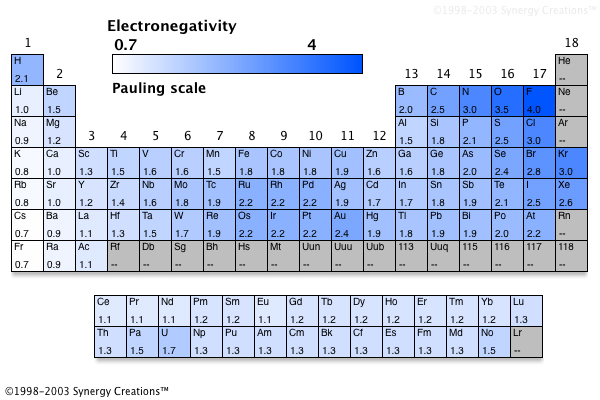
Photo: FrozenMan at English Wikipedia, CC0, Public Domain The atom with the greater value will basically take the electron bond from the other atom and possess it, creating an ionic bond.Ī visualization of a molecule’s electrostatic potential. If two atoms have electronegativity values that are extremely different, they won’t share electrons between them at all. Though sometimes two atoms will have the exact same electronegativity values and have a covalent bond, meaning that they equally share the electrons. In most cases, the electrons found within a chemical bond have a greater attraction to one atom than to the other atom, which creates a polar covalent bond. What is electronegativity exactly? Electronegativity refers to an atom’s ability to attract the electrons present in a chemical bond, or an atom’s ability to attract electrons when that atom is part of a specific compound. What Is An Electronegativity Trend?īefore we can begin looking at examples of the electronegativity trend, let’s define our terms. While this is the basic definition of the electronegativity trend, to truly understand it, it would be helpful to put it in perspective and look at some specific examples of the trend. This trend is seen as you move across the periodic table from left to right: the electronegativity increases while it decreases as you move down a group of elements. The electronegativity trend refers to a trend that can be seen across the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
